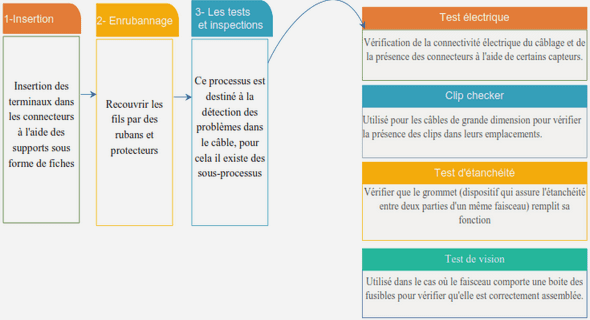
MD Simulations Combined NMR SAXS Data Characterizing IDP Conformational
Ensembles
NMR and SAXS data for validating MD simulations
Toward force fields for IPDs In the late 20th century, the most popular all-atom force fields for proteins were GROMOS96 [354], OPLS-AA [166], CHARMM22 [211], or AMBER99 [362] (Fig. 2.2). These force fields were optimized to well reproduce the structure and dynamics of folded proteins, such as enzyme and receptors, in explicit solvent, using generally a three-site water model, such as the SPC/E [18] or TIP3P [165]. However, with the emergence of IDPs in structural biology, limitations in accuracy of modeling protein disordered regions appeared at the beginning of the 2000s.
More specifically, it was pointed out that MD simulations with traditional force fields often generated conformational ensembles of disordered segments with over-estimated propensities to form secondary structures and overly compact shapes when compared to experimental data. Since then, great efforts were made to develop new force fields optimized for both structured and disordered parts of proteins. In this section, we survey these developments which have been reported in a about fifteen publications since 2001. Early efforts to optimize force fields were focused on improving the backbone φ and ψ dihedral potentials to better reproduce the two-dimensional Ramachandran probability distributions, notably resulting in the refined force fields OPLS-AA/L [167] and AMBER03 [85].
More explicitly, MacKerell et al. introduced in CHARMM22 cross term energy functions of φ and ψ angles to better account for their correlations. These grid-based energy correction maps (CMAP) gave the name CHARMM22-CMAP to this new force field, also referred to as CHARMM27 [212, 213]. Since, many improvements of the backbone φ and ψ dihedral energy potentials were continuously made, leading to a serie of force fields more or less able to well reproduce polypeptide propensities to form local secondary structures. This includes the AMBER99SB [150], AMBER99SB∗ , AMBER03∗ [23], AMBER03w [24], CHARMM22∗ [266], CHARMM36 [151], and AMBER-14SB [214] force fields (Fig. 2.2).
It could be noted that some of these 26 2.1. NMR and SAXS data for validating MD simulations models were subsequently completed with improved sides chain torsion potentials to refine side chain rotamers, resulting in the « -ILDN » extension of some force field names, such as AMBER-99SB-ILDN [202]. More recently, updated CMAP potentials were implemented in several force fields to specifically correct the φ and ψ probability distributions of intrinsically disordered proteins, leading to the new force fields . From a methodological viewpoint, one should recall that all these developments were mainly validated with comparisons between back-calculated and experimentally measured NMR observables, such as scalar 3J-couplings or residual dipolar couplings (RDCs), on both folded and unstructured polypeptides. Most recent ones, such as CHARMM36m, also used SAXS data for additional validations.
Force field benchmark using NMR and/or SAXS data
Over the past four years, several benchmarks of previously mentioned force fields were performed on various disordered peptides in order to guide the choice of the most appropriate ones for IDP simulations. The IDPs which frequently served as test cases are amyloidogenic peptides, particularly Aβ40 and Aβ42 related to the Alzheimer’s disease, or the 37-residue IAPP involved in the type II diabetes. These disordered peptides are known to have a significant propensity to form transient β-strands accountable for their aggregation and fibrillization. When the force field capability to reproduce their secondary structure propensities is tested against NMR measurements of chemical shifts, 3J-couplings, or residual dipolar couplings, then a general trend seems to emerge :
AMBER03 and CHARMM22 are not appropriate for this kind of β-peptides since they over-stabilize α-helices, while OPLS-AA and GROMOS96 similarly produces unstructured ensembles with low secondary structures propensities [126, 147, 326]. In contrast, AMBER99SB, AMBER99SB-ILDN, AMBER99SB∗ -ILDN, and AMBER03w provides similar results in good agreement with NMR data, and CHARMM22∗ slightly performs better than all the previously mentioned force fields [42, 147, 326]. This hierarchy is overall confirmed by other studies on shorter peptides of about 10 to 20 residues [56, 257]. Interestingly, AMBER99SB-ILDN and AMBER99SB∗ -ILDN were also shown to produce secondary structure propensities consistent with NMR data for disordered peptides with α-helix tendencies, such as the central region (380-490) of axin-1 protein [28] or the TAD2 peptide (41-62) from the human p53 tumor suppressor.