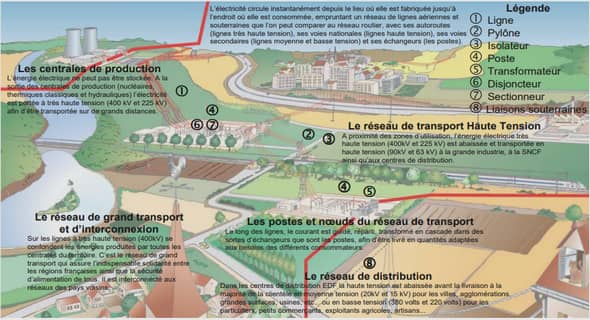
Cours geologic and geographic context, tutoriel & guide de travaux pratiques en pdf.
Regional Piezometry
In terms of regional groundwater flow a semi-regional groundwater divide line can be drawn across the Appalachian Highlands (Figure 4). Because of its prominent elevation as well as its limited or negligible surficial sediment cover, the Appalachian region can be considered a recharge area while the St. Lawrence River, marking the northern boundary and the lowest point of the study area, represents the regional flow outlet (Figures 4 and 5). Streams and rivers within the numerous sub-basins also act as local discharge areas, particularly since the highly fragmented terrain of the Appalachians forms a dense hydraulic network (Figure 4). The extent and contribution of regional flow to the basin-scale water budget is not yet well defined. However, the conceptual groundwater flow system of the Chaudière-Appalaches region appears to fit Tóth’s (1963) concept of nested groundwater flow, in which local flow regimes are embedded within larger intermediate or regional flow regimes (Figure 5).
Figure 4 Water table elevation with respect to average sea level (m), modified from Lefebvre et al. (2015)
Figure 5 North-south regional cross-Section of the study area showing conceptual model of regional flow
Total precipitation over the study area is estimated to be 1149 mm/year, with an average of 166 mm/year of recharge reaching the fractured rock aquifer (Lefebvre et al. 2015). Spatial distribution of recharge is, however, highly variable. In the St. Lawrence Lowlands, areas covered with thick layers of fine sediments can receive less than 15 mm/year whereas rock outcrop areas can receive more than 300 mm/year. In the Appalachian Highlands, where the surficial sediments are fairly thin and permeable, aquifer recharge tends to be higher, varying from 100 mm/year to more than 300 mm/year. Additional studies on specific watersheds within the study area have reported overall recharge rates to the bedrock aquifer of between 27 mm/year and 38 mm/year (Benoît et al. 2014 and L’Heureux 2005, respectively). Discrepancies between recharge values could be attributed to the spatial variability of the bedrock recharge.
Geochemical Setting
Major Ion Chemistry
The chemical composition of a groundwater sample holds a remarkable amount of information on the path taken by groundwater (Appelo & Postma 2004; Clark and Fritz 1997; Hounslow 1995). As recharge water enters the subsurface and flows downgradient, its interactions with the minerals, gases and, to some extent, any living organisms along its path, will change the water’s chemical composition (Tóth 1999; Cloutier et al. 2008).
Benoît et al. (2014) and Lefebvre et al. (2015) carried out a thorough analysis of the regional groundwater geochemistry for the Chaudière River watershed and the Chaudière-Appalaches region, respectively. The first 155 groundwater samples analysed by Benoît et al. (2014) were included in the subsequent geochemical analysis performed by Lefebvre et al. (2015) thus only the major ion water groups defined in the second study are discussed in this Section. Lefebvre et al. (2015) collected an additional 200 samples and classified a total of 355 water samples, collected mostly from residential wells (approximately 60 m deep) across the study area, into seven geochemical composition groups based on the relative proportions of their major ions. From the seven groups, three were considered as resulting from anthropogenic contamination and four were retained as natural water types, one of which is present only in a small part of the study area and thus was not included in the regional geochemical analysis (Figure 6).
The three main natural water types identified in the study area are classified as follows:
Group 1 (G1): Water of type Ca-HCO3, generally associated with young recharge waters.
Group 2 (G2): Water of type Na-HCO3, typically evolved from Ca-HCO3 waters which have undergone ion exchange in the subsurface in which Ca2+ ions were exchanged for Na+ ions.
Group 3 (G3): Water of type Na-HCO3/Na-Cl, considered as highly evolved. High Na+ and low Ca2+ concentrations result from ion exchange and an elevated Cl- concentration is likely to be associated with the dissolution of salts. The origins of the chloride ions are discussed in later Sections.
For reference and comparison, four additional water types related to the study site were added to the Piper diagram: 1) the composition of a typical seawater sample (Hem 1985), 2) the composition of Sample S77 considered as Champlain Sea seawater (Cloutier et al. 2012), 3) a sample from the deepest observation well over the study area (Bordeleau et al.
2015), as well as 4) the composition of brines collected in the Cambrian-Ordovician sedimentary formation lying below the Utica Shale in the Bécancour area located to the west of the study area (Pinti et al. 2011).
Figure 6 Piper diagram of natural water types found in the study area
The spatial distribution of the major ion water groups is consistent with the conceptual flow model discussed earlier (Figure 5). Water from group G1, associated with recharge water, can be found across the study area but is primarily found in the Appalachian Highlands, a region presumably characterized by local flow systems (supported by the geochemistry) which would imply rapid transit times. On the other hand, the dominant water type across the St. Lawrence Lowlands is water from the more evolved group G3, implying that these samples would have had time to undergo geochemical changes. This suggests that the sampled groundwater in the St. Lawrence Lowlands has likely traveled over a longer distance and is relatively older. Water in group G2, which has undergone some level of ion exchange, is also mostly found in the St. Lawrence Lowlands. Travel times for this water group are expected to be longer than those of group 1 and shorter than those of G3.
Evidence of Geochemical Processes
Major ion analysis of waters across the study area identified three dominant geochemical processes in the region: carbonate dissolution, ion exchange and mixing. Moreover, the proposed conceptual groundwater flow model suggests a potential input of deep fossil brines to near-surface waters, supporting the hypothesis of mixing of fresh waters and brines.
Dissolution of carbonates
Mineral dissolution is controlled by the availability of the dissolving minerals (reactants) and the solubility of the mineral (reaction rate) (Freeze and Cherry, 1979). Since the most soluble minerals, such as halite, can easily be leached away, areas with rapid renewal of water, such as recharge areas, are generally left with the slower dissolving minerals such as carbonates (Freeze and Cherry, 1979). For the present study, carbonate minerals are of particular interest since they not only have a low solubility but they are also present in the study area as 1) the main building blocks of the sedimentary St. Lawrence Platform, 2) components of the till overburden resulting from the movement of glaciers from the St. Lawrence Lowlands towards the Appalachians and 3) to a certain extent, present as cementing mineral in the rocks and fractures of the Appalachian rock formations.
As noted by Freeze and Cherry (1979), only a small or insignificant amount of carbonate minerals can be sufficient to create saturated conditions and influence groundwater chemistry. Thus, across our study area, carbonate dissolution is inevitably taking place early along the flow path; during and soon after recharge. This is indicated by the presence of G1 water, with major ions Ca2+ and HCO3, signifying that dissolution of carbonate minerals has occurred through either the surficial sediments or shallow bedrock.
The equilibrium dissolution-precipitation reactions of the two most common carbonate minerals, calcite and dolomite, are given as follows:
Calcite: CaCO3Ca2++ CO32- (1)
Dolomite: CaMg(CO3)2 Ca2+ + Mg2+ + 2CO32- (2)
When water enters the subsurface it first migrates through the unsaturated soil zone, where groundwater equilibrates with carbon dioxide (given off through root respiration and the decay of organic matter) to pCO2 levels that are higher than that of the atmosphere (Freeze and Cherry, 1979; Appelo and Postma 2005). Appelo and Postma (2005) point to pCO2 levels in the active soil zone ranging from the atmospheric value of 10-3.5 atm to as high as 10-1.5 atm. Dissolution of gaseous CO2 into water produces excess H+ ions, which at a pH of less than 8.3, bind to CO32- ions to produce HCO3- which drives the dissolution of calcite and dolomite (Freeze and Cherry, 1979). The equilibrium dissolution-precipitation reactions then become:
Calcite: CO2(g) + H2O + CaCO3 Ca2+ + 2HCO3- (3)
Dolomite: 2CO2(g) + 2H2O + CaMg(CO3)2 Ca2+ + Mg2+ + 4HCO3- (4)
Water is considered to be in an open system when it is in equilibrium with “soil zone” CO2. In this case, pCO2 stays constant while carbonate minerals dissolve until equilibrium is reached. However, if the system is closed and therefore not receiving CO2 input from the soil zone, pCO2 will decrease until equilibrium is reached.
In Figure 7.A the carbon, calcite and dolomite equilibrium lines represent a pure water sample at an average temperature of 9.6 oC equilibrated using PHREEQC version 3 (Parkhurst and Appelo 1999) with CO2(g), CO2(g) and calcite, and CO2(g) and dolomite, respectively. Arrows in Figure 7 show the dissolution path in open and closed systems. The distribution shows that most samples from G1 have not yet reached calcite and dolomite equilibrium while samples from G2 and G3 appear to be saturated or supersaturated with respect to dolomite.
The final control on the dissolution of carbonate minerals is not their availability but their solubility which, for calcite and dolomite, is inversely dependant on temperature; dolomite becoming progressively more soluble than calcite with decreasing temperature (Szramek et al. 2007). At a temperature of approximately 10 oC, because of the relative solubility between the minerals, calcite might reach equilibrium and become supersaturated while dolomite remains undersaturated (Szramek et al. 2007). Furthermore, Freeze and Cherry (1979) state that in North America, supersaturation can result from the temperature gradient in the soil during spring. During spring recharge, dissolution occurs at low temperatures under open conditions. But, at a few meters of depth, temperatures can be several degrees higher than at the surface, leaving the water supersaturated in calcite and dolomite. The temperature difference between the recharge zone and the aquifer could explain the calcite supersaturation of water samples from G2 and G3 observed in Figures 7.A, 7.C and 7.D.
Figures 7.A and 7.C also confirm that by having pCO2 levels lower than atmospheric levels, all samples from G3 were likely collected from closed system conditions in which the carbon dioxide initially present in the system was consumed by the dissolution of carbonates until equilibrium. Conversely, samples from G1 and G2 have pCO2 levels corresponding to that of the soil zone, thus indicating that these samples either reached equilibrium under open conditions or that pCO2 in the soil zone was initially high.
Figure 7 Evidence of carbonate dissolution in the C-A study area: a) Calcite and dolomite equilibrium with respect to pH and partial pressure of CO2; b) Ca2+ and Mg2+ ratios of dissolving carbonates, c) Saturation of calcite with respect to partial pressure of CO2, and d) Saturation of calcite with respect to dolomite.
Freeze and Cherry (1979) state that despite laboratory experiments showing that calcite equilibrium can be reached within hours or days, field rates are often slower. Nevertheless, carbonate dissolution reactions can be considered as being fairly rapid over a regional-scale flow system. Thus, samples that have not reached calcite and dolomite equilibrium, mainly those from G1, can be considered as being recently recharged water. Furthermore, high pCO2 levels can also indicate that the aquifer has a shallow circulation zone keeping the system in open conditions or that the flow distance between recharge and discharge zones is short. The large range of pCO2 and pH across the samples testifies to the variability of recharge conditions and state of evolution among the water samples.
In this context, the low final pCO2 levels of waters from G3 relative to G1 could indicate that G3 waters were recharged under lower initial soil pCO2 levels.
For a system in which the only carbon sources are dissolved carbon dioxide and carbonate minerals, stoichiometry can be used to derive the expected ratios between the concentrations of dissolved inorganic carbon (DIC) and Ca2+ ions for minerals dissolving in various proportions (Figure 8). The data show that water from group G1 has DIC/Ca2+ ratios corresponding to the dissolution of calcite and dolomite, while samples from G2 and G3 are affected by a process removing Ca2+ ions from the system (Figure 9a).
Cation exchange
Cloutier et al. (2010), Lefebvre et al. (2015) and Benoît et al. (2014) identified Ca2+/Na+ ion exchange as another important geochemical process in the study area. Ion exchange is described as the process by which one ion is replaced by another ion at the surface of a solid (Appelo and Postma 2005). In Chaudière Appalaches, this reaction can be described through the following equation: ½ Ca2++ Na+-X½ Ca2+-X + Na+ (5)
In this case, when considering the Na+ ions as having been exchanged for Ca2+ (Figure 8B), water samples from groups G2 and G3 appear to be strongly affected by the cation exchange process (Figures 9A and 8B).
As a side note, it is worth noting that when accounting for ion exchange, the ratio of dissolved dolomite to calcite indicates that calcite is the main dissolving mineral and that water from G3 is affected by yet another process adding Na+ ions to the system (Figure 8B).